|
6/18/2023 0 Comments Example of electron capture from the nuclear shell of the electron Elementary particle with a negative electrical elementary ch. The gas in the detector chamber is ionized by the radiation particles. Some examples of radioactive isotopes used are 3 H, 63 Ni, 85 Kr, and 90 Sr. When a nuclear change takes place, an atom changes its identity and is now an atom of a different element. This is one of the means that a nuclear change can take place. capture capture of an electron Elementary particle with a negative electrical elementary ch. Electron capture ionization is the ionization of a gas phase atom or molecule by attachment of an electron to create an ion of the form. Thanks so much for your question regarding electron capture. Example: K-40 + e- => Ar-40.Įlectron Elementary particle with a negative electrical elementary ch. which is smaller by one unit the mass number Mass of an atom in nuclear mass units. has an atomic number Number of protons in an atomic nucleus. The formed nuclide A nuclide is a type of atom characterized by its proton numb. Confirming a decades-old prediction, scientists found the first clear example of a type of stellar explosion called an electron-capture supernova (large white dot at right). 
converts into a neutron Uncharged elementary particle with a mass of 1.67492716 ♱0. in the nucleus Positively charged nucleus of an atom. of the atomic shell, whereby a proton Elementary particle with a positive electrical elementary ch. captures an electron Elementary particle with a negative electrical elementary ch. of an atom Smallest particle of an element which cannot be chemically d. Electron capture is a process in which a parent nucleus captures one of its orbital electrons and emits a neutrino. The nucleus Positively charged nucleus of an atom. Disintegration type of some radionuclides, e.g. Of the three modes, this invention relates about the phenomenon of electron capture but not through the decay process of an atom. History and etymologyĮxperimental confirmation of electron capture was first reported by Luis Alvarez in 1936 for Vanadium-48. The significance of electron capture decay pathway being that no electrons are emitted and therefore reduced risk to adjacent tissue. Medical ApplicationsĪn isotope making use of electron decay is Iodine-123 as a tracer in thyroid imaging which decays by electron capture to Tellurium-123 which emits a low energy gamma ray for detection. For this example we have used only single-centered basis states that are (linear combinations of) eigenstates of the unperturbed target Hamiltonian. Confirming a decades-old prediction, scientists found the first clear example of a type of stellar explosion called an electron-capture supernova (large white dot at right). Typically, it occurs in heavy nuclei with a relative abundance of protons or where insufficient decay energy exists for positron emission to occur. These transitions may result in the loss of an outer orbital electron, where it is ejected as an Auger electron.Įlectron capture is rarely an exclusive decay mode and typically occurs alongside beta-plus decay within the radioactive sample. 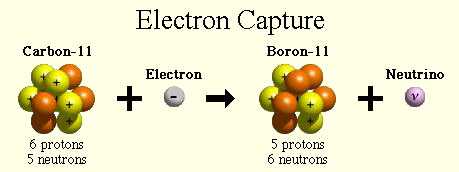
The vacancy left by this electron is then filled by an outer orbital electron setting off a cascade of electron transitions with their associated characteristic x-rays emission. A continuous spectra of Bremsstrahlung is radiated as the inner shell electron is propelled towards the nucleus. The best-known example is of potassium 40 : 11 of the nuclei of that isotope of. The electron capture decay pathway is usually associated with several other processes. Electron capture is a comparatively minor decay mode caused by the weak force. The decay energy is almost wholly transferred to the emitted neutrino with a characteristic quanta of energy. The decay pathway has similarities to beta decay and is often termed inverse beta decay . Note the reduction in atomic number but conservation of mass number in the daughter nucleus. In electron capture, an electron from an inner orbital is captured by the nucleus of the atom and combined with a proton to form a neutron. The electron on the left side of the equation is usually absorbed from the K or L shell of the parent nucleus. 
The nuclear reaction depicting electron capture decay is: Accompanying this decay method is the emission of Bremsstrahlung, characteristic x-ray emission, gamma rays and Auger electrons. As with the beta plus decay the atom changes to a different element but with the same number of particles in the nucleus. Electron capture is the radioactive decay process by which an atom's inner orbital electron is absorbed within the nucleus followed by conversion of a proton to a neutron and emission of a neutrino (ve). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
